SPECTROSCOPY: AFM improves resolution of IR spectroscopy
CRAIG PRATER, KEVIN KJOLLER, ROSHAN SHETTY, and ALEXANDRE DAZZI
The atomic force microscope (AFM) has been enormously successful addressing nanoscale measurement problems in basic research, materials science, and engineering. In fact, the AFM is credited with enabling multibillion-dollar research investments in nanoscience and nanotechnology because it provided researchers with one of the first tools to visualize and measure complex structures with nanoscale spatial resolution. A significant gap in AFM capabilities, however, is the technique's inability to chemically characterize a sample. The AFM can measure mechanical, electrical, magnetic, and thermal properties of a material, but on its own, AFM lacks the robust ability to chemically characterize materials.
In contrast, infrared (IR) spectroscopy is widely used to chemically characterize countless materials, but its spatial resolution is restricted by the diffraction limit. In practical terms, the resolution of transmission Fourier transform (FT) IR spectroscopy is limited to about three times the wavelength of the IR radiation, and even attenuated total reflectance (ATR) spectroscopy can only achieve resolution approaching the source wavelength.
Although there have been some encouraging attempts to combine AFM with optical techniques, until recently they have been limited in wavelength range or spatial resolution. A new technique, AFM-based infrared spectroscopy (AFM-IR) has emerged that successfully combines the nanoscale spatial resolution capabilities of AFM with IR spectroscopy. The result is an instrument that is able to measure and map local chemical composition below the diffraction limit, as well as perform nanoscale topographic, mechanical, and thermal analysis.
nanoIR
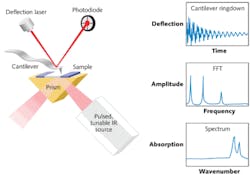
Originally developed by Alexandre Dazzi and coworkers in France, the AFM-IR technique has recently been commercialized by Anasys Instruments.1 Named nanoIR, the instrument uses a pulsed, tunable IR laser source based on an optical parametric oscillator (OPO) to excite molecular vibrations in a sample that has been previously mounted to an IR transparent zinc selenide prism (see Fig. 1). The IR beam illuminates the sample by total internal reflection similar to conventional ATR spectroscopy. As the sample absorbs radiation it heats up, leading to rapid thermal expansion that excites resonant oscillations of the AFM's cantilever. These induced oscillations decay in a characteristic ringdown pattern.
The ringdown pattern can be analyzed using Fourier techniques to extract the amplitudes and frequencies of the oscillations. The amplitudes of the cantilever ringdown as a function of the source wavelength provide local absorption spectra, while the oscillation frequencies are related to the mechanical stiffness of the sample. The IR source can also be tuned to a single wavelength to simultaneously map surface topography, mechanical properties, and IR absorption in selected absorption bands.
Capabilities
The nanoIR instrument enables a suite of critical measurement capabilities that were previously unavailable by overcoming the resolution limits of IR spectroscopy and adding chemical and mechanical mapping to the AFM. The instrument can make detailed studies of the structural and chemical makeup of complex samples and can be applied to characterize polymer blends, multilayer films, coatings, and other materials. Modern polymer blends can contain many different components, materials for strength, shock absorption, electrical conductivity, flame retardants, and compatibilizers, for example. The technique also has applications in the life sciences. The new technology enables users to quickly survey regions of a sample via AFM and then rapidly acquire high-resolution chemical spectra at selected regions on the sample, allowing quick identification of sub-wavelength domains. Making additional mechanical and thermal property measurements with nanoscale resolution adds significantly to the power of this technique and enables structure/chemistry/property correlations.
The nanoIR system enables researchers to harness the full power of mid-IR spectroscopy. The system's IR OPO source is continuously tunable from 3600 to 1200 cm-1. This range covers a major portion of the mid-IR, including important CH, NH, and CO bands, as well as carbonyl and amide I/II bands. Polymer spectra acquired with the nanoIR have demonstrated good correlation with bulk FTIR spectra. The system software also allows researchers to export absorption spectra to standard analysis packages to rapidly analyze samples and identify chemical components.
In addition to its ability to provide high-resolution IR spectra, the software allows integrated thermal and mechanical property mapping. Mechanical properties of a sample can be collected using a contact resonance method to map stiffness variations simultaneously with the topography and chemical information. The platform can also perform nanoscale thermal analysis, allowing researchers to work beyond bulk thermal analysis measurements to obtain information not available with any other technique.Applications
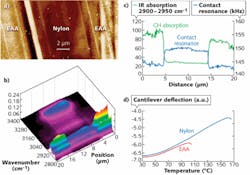
Two examples illustrate the power and utility of AFM-IR. In one, the nanoIR instrument maps layers and interfaces in multilayer films, including layers that are too thin to resolve with conventional IR microspectroscopy (see Fig. 2).2 The other example involves in situ identification and imaging of polymer nanogranules in bacteria.3 The work looked at Rhodobacter capsulatus individual bacteria and the tracking of energy storing the polymer polyhydroxybutyrate (PHB). Structures were identified in the size range of 50 to 400 nm, illustrating the potential of the technique (see Fig. 3).While it was not difficult to imagine that there would be many applications for AFM-IR in the "nanospace" of the polymer world, it is encouraging to see this technique extending into the world of biology. The work of Dazzi and others around the world in applying AFM-IR to life sciences research highlights the utility of spatially resolved chemical analysis in life sciences at levels previously not believed possible.
REFERENCES
1. A. Dazzi, R. Prazeres, F. Glotin et al., "Chemical Mapping of the Distribution of viruses into infected bacteria with a photothermal method," Ultramicroscopy (2007).
2. C. Prater, K. Kjoller, D. Cook, G. Meyers et al., "Nanoscale IR Spectroscopy of materials using an AFM," Microscopy & Analysis (April 2010).
3. C. Mayet, A. Dazzi, R. Prazeres, J.M. Ortega, and D. Jaillard, "In situ identification and imaging of bacterial polymer nanogranules by infrared nanospectroscopy," Analyst 2010; doi: 10.1039/C0AN00290A.
FURTHER READING
A. Dazzi et al., "Sub-100nm spectromicroscopy of living cells," Opt. Lett., 13, 14 (July 15, 2008).
A. Dazzi, F. Glotin, and R. Carminati, "Theory of infrared nano-spectroscopy by PhotoThermal Induced Resonance," J. Appl. Phys., 107, 124519 (2010); doi:10.1063/1.3429214.
K. Kjoller, J. Felts, D. Cook, C. Prater, and W. King, "High-sensitivity nanometer-scale infrared spectroscopy using a contact mode microcantilever with an internal resonator," Nanotechnol., 21, 185705 (2010).
C. Policar et al., "Subcellular IR Imaging of a Metal-Carbonyl Moiety Using Photothermally Induced Resonance," Angewandte Chemie International Edition; doi: 10.1002/anie.201003161.
A. Dazzi, chapter on AFMIR technique and applications, in Thermal Nanosystems and Nanomaterials, Springer (2009).
J. Houel, E. Homeyer, S. Sauvage, P. Boucaud, A. Dazzi et al., "Mid-IR absorption measured at Lambda/400 resolution with AFM," Opt. Exp., 17, 13, 10887-10894 (2009).
A. Dazzi, chapter on AFMIR technique and applications, in Biomedical Vibrational Spectroscopy, P. Lasch and J. Kneipp, ed., Wiley (April 2008).
A. Dazzi, R. Prazeres, F. Glotin, and M. Ortega, "Subwavelength infrared spectromicroscopy using an AFM as a local absorption sensor," Infrared Phys. and Technol., 49, 113-121 (2006).
Craig Prater is CTO, Kevin Kjoller is VP, product development, and Roshan Shetty is CEO at Anasys Instruments, 121 Gray Ave., Suite 100, Santa Barbara, CA 93101. Alexandre Dazzi is an associate professor at Université Paris-Sud, Orsay, France. E-mail: [email protected]; www.anasysinstruments.com.