SPECTROSCOPY: Diode source provides high resolution at UV wavelengths
To measure concentrations of pollutants and greenhouse gases outside of a laboratory, researchers wish for a device that is small, robust, gathers data quickly, runs on battery power, and is sensitive and specific. An experimental ultraviolet (UV) spectroscopy system that comes close to fulfilling that wish has been built by collaborators at the Naval Research Laboratory (NRL; Washington, DC) and Sandia National Laboratories (Livermore, CA). Jeffrey Koplow, Dahv Kliner, and Lew Goldberg built a source for UV-absorption spectroscopy using a frequency-quadrupled diode-laser-based system. The instrument measured the spectrum of sulfur dioxide (SO2) with considerably higher resolution than has previously been reported.
Compact infrared (IR) absorption spectroscopy systems have been developed using diode lasers, but UV spectroscopy offers some specific advantages; in the UV, many small molecules have larger absorption cross sections, and neither sunlight nor water vapor interferes with the measurement. Another major benefit of UV spectroscopy is that inexpensive silicon detectors with very little noise work at room temperature, unlike IR detectors that often require cooling. Comments Kliner, "UV detectors can be extraordinarily sensitive, permitting single-photon counting."
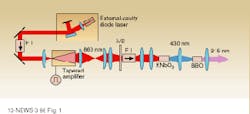
More than 5 mW of single-longitudinal-mode light from an external-cavity diode laser seeds an aluminum gallium arsenide (AlGaAs) tapered amplifier (see Fig. 1). The tapered amplifier, developed by SDL (San Jose, CA), provides relatively high power while maintaining good beam quality. The amplifier is pulsed to provide peak output powers of 11 W. The amplified beam is frequency-doubled with potassium niobate to 430 nm, then quadrupled to 215 nm with beta barium borate (BBO).
Wavelength tuning is provided by tuning the seed laser from 842 to 868 nm. Pulsed operation, provided by pulsing the current to the tapered amplifier, provides more efficient frequency conversion. While operating in the single-mode configuration, the laser provides only 40 nW of UV output, which is sufficient for UV-absorption spectroscopy using off-the-shelf silicon photodiodes. More important, the system provides an unusually narrow linewidth in the UV of less than 200 MHz. In comparison, frequency-doubled or tripled dye lasers—conventional sources for UV spectroscopy—often have linewidths of several gigahertz.
Unique advantages
Dye lasers have several drawbacks for field use, including complexity, large size, and weight. In the last several years, frequency-converted optical parametric oscillators (OPOs), which reduce the amount of plumbing necessary, have also been developed for this application. The OPOs, however, tend to consume a lot of power, and most are relatively broadband sources.
Diode-laser-based systems have the potential to be small and simple. Although, in this case, the researchers were not trying to minimize the size, the system could be engineered to fit onto a 1-sq ft breadboard. With folding mirrors, the UV laser system could shrink to the size of a shoebox.
Several other groups of researchers have been developing diode-based UV sources. The NRL/Sandia group`s work stands out, though, for being deeper into the UV—most other groups work at wavelengths longer than 300 nm—and for combining tunability with narrow bandwidth.
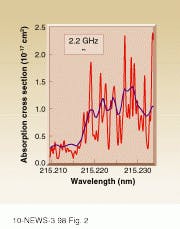
Kliner and his coworkers demonstrated the instrument`s capabilities by measuring the UV spectra of nitric oxide and SO2. Scientists studying pollution and climate are interested in both of these gases. Nitric oxide plays a central role in ozone chemistry, while sulfur dioxide is the source of both acid rain and sulfate aerosols in the atmosphere. The spectrum for sulfur dioxide had considerably finer resolution than previously published spectra (see Fig. 2). Kliner reproduced and verified the spectrum using a dye laser.Looking forward
The next steps for the researchers include increasing the UV power and optimizing the pulse length. Increased output power would be useful for other types of UV spectroscopy, including laser-induced fluorescence. Other research groups have produced more power by putting the nonlinear crystals into resonant cavities, but for now Kliner and his coworkers are avoiding this tactic in favor of the simplicity of single-pass system. Instead, they are considering using larger amplifiers and shortening the pulse duration.
The tapered amplifiers appear to work better when the pulses are short—in a previous experiment the system produced three orders of magnitude more output power (15 µW) in 17-ps pulses. Such short pulses dictate broadened bandwidth, however. The researchers believe that pulses about 1 ns long will provide sufficiently narrow bandwidth and will still be fast enough to make the tapered amplifiers work efficiently.
The researchers discovered that, as pump powers increase, the tapered amplifiers begin to degrade, and the first evidence of this is in reduced beam quality. Then the power output drops, and eventually the device experiences catastrophic failure. Kliner comments, "We blew out several" of the $6000/chip amplifiers during the study. He expects that the group will perform further work on tapered-amplifier life expectancy. Commenting on the cost of the tapered amplifier as part of the overall system cost, Kliner notes, "It’s expensive for a diode laser, but cheap for a UV spectroscopy laser."