FIBER-OPTIC SENSORS: Optics breathe new life into oxygen sensing
Advances in high-performance sensor materials and optoelectronics have enabled novel optical sensors for use in a diverse array of markets including the life sciences, environmental, food and beverage, process control, and aviation. Compared to traditional electrochemical sensing techniques such as galvanic, paramagnetic, and fuel cell sensors, these optical sensors (sometimes called "optrodes," as in optical electrodes) can be made in small and customizable form factors. They also have faster response, provide long-term calibration free stability, are chemically inert, and couple easily to optical fibers for remote measurements (see table).
The principle of operation for optrodes is to trap a target-sensitive fluorophore or pH indicator dye in a host matrix that can then be applied to the tip of a fiber, flat substrate, adhesive membrane, or other surface. Optical fibers are particularly versatile because they can be made into probes of many sizes and shapes and can be jacketed for deployment in harsh environments. The sensing portion of the fiber probe is the tip, which can be made in sizes from 50 to more than 1000 µm in diameter.
Oxygen sensor
Oxygen optrodes are relatively new compared to electrochemical sensors. As chemistries and detector electronics improve the use of optrode sensors is increasing, especially in the life-sciences and food-packaging markets. Most optrodes use a polymer-based host matrix to trap the fluorophore. Polymer-based oxygen (O2) optrodes have the advantage that they can be produced in volume with excellent lot-to-lot repeatability for calibration consistency. The main disadvantage is that the electronic environment of the polymers promotes photodegradation of the fluorophore, so the overall operating lifetime is limited.
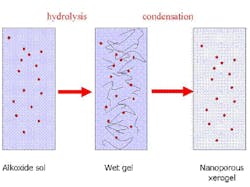
Another chemistry first pioneered as a host matrix for O2 sensing in the 1990s uses sol gels (see Fig. 1).1, 2 Sol gels are a nanoporous, glass-like material that typically consist of alkoxide metal molecules that have been mixed with water and solvent to create a homogeneous partially polymerized liquid called a sol. The fluorophore is dissolved in the sol that is then applied to the substrate host (fiber tip or flat substrate) as a thin film, dried and cured to a hardened glass-like substance. Sol gels are widely used in many manufactured products as protective coatings, and are also easy to manufacture in high volume.
Sol gels offer some unique advantages compared to polymer technology. These include superior bonding to a wide range of substrates--they do not chemically bond to the sensor indicator, but instead "cage" or physically encapsulate the molecule, which provides better stability of the indicator molecule and makes the sol gel more versatile in terms of the range of indicators that can be trapped. The sol-gel polarity and porosity can be tailored by changes in recipe and curing details to match the requirements of the target sample chemistry. So, for example, pH sol gels are made hydrophilic, O2 sol gels are made hydrophobic, and for use in hydrocarbons like jet fuels, they are made both hydrophobic and lipophobic (repels long chain hydrocarbons).
In addition, sol gels exhibit superior optical properties—they can be made transparent, and readily transmit light from the ultraviolet through the near-infrared. Sol gels can be combined with polymers in multilayer probes--so, for example, a sol-gel O2 optrode can resist photobleaching, but can be protected from harsh chemicals by an overcoat of silicone. And available sol-gel probe chemistries include standard materials that are used for gases and aqueous samples. These chemistries are used widely for life-science applications, environmental monitoring, and other relatively benign gas and liquid samples. A lipophobic, hydrocarbon-resistant formulation is used for compatibility with oils, alcohols, and hydrocarbon based liquids and vapors. This has been tested successfully in aviation fuels, gasoline, diesel fuel, hydraulic fluids, and wines. The sol gels can be used with a ruthenium-based fluorophore for the 0% to 100% oxygen range. A platinum-poryphrin fluorophore is used for trace amounts of O2 in the 0% to 5% range. Sensitivity of this sensor is in the low parts per million of O2 in gas, and low parts per billion in liquids.
Advantages of the oxygen optrode
For oxygen monitoring, the fiber optic and sensor tip are illuminated with a blue LED creating a red luminescence (see Fig. 2). Luminescence generated at the tip is collected by the probe and carried by the optical fiber to a phase fluorometer. When O2 in the gas or liquid sample diffuses into the thin-film coating, it dynamically quenches the fluorescence.
The degree of quenching correlates to the collision frequency between O2 molecules and the excited state of the fluorophore, and is therefore directly reactive to the partial pressure of O2. The quenching is observed as a decrease in the average fluorescence lifetime, which is detected as a phase shift between the fluorescent signal and a pulsed blue LED.
The change in phase (or lifetime or intensity) and p O2 are related by the Stern-Volmer equation. Probes are calibrated by exposure to known levels of p O2 and fitted with linear Stern-Volmer plots or polynomials for extended ranges. Temperature and total pressure compensation calibrations are determined in a similar fashion.
Revolutionary pH technology
Advances in the sol-gel chemistry have led to the development of a unique hydrophilic fiber-optic pH sensor offering the same fiber and probe capabilities as the oxygen optrode.3 The sensor is based on measuring the ratio of absorbance of a pH indicator dye at two wavelengths. The ratiometric method is immune to changes in intensity and moderate changes in dye concentration due to leaching, plus it is easy to calibrate.
Calibration consists of exposing the optrode to standard buffer solutions. Different pH indicators may be trapped in the sol gel for use in different pH ranges. The precision of the sensor is limited by the precision of the spectrophotometer and the total absorbance of the sensor. For an indicator with an absorbance of 1, and a spectrophotometer with a precision of 0.001 AU, a pH precision of approximately 0.001 pH units is possible.
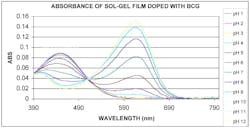
The bromocresol green (BCG) optrode is responsive from pH 5 to 9, ideal for biological systems, environmental sampling, and many industrial processes (see Fig. 3). The phenol red optrode has a low pH range of about 1 to 3 and a high range of about 9 to 12. Other indicator dyes and sol-gel chemistries are possible for acidic and basic pH ranges.
Visual-based colorimetric pH devices were made for the aquarium industry in the late 1980s. Ocean Optics pioneered the transfer of this technology for use as optrodes in the early 1990s. The material was based on polymer films that trapped the indicator molecules. These sensors were slow and the indicator gradually leached out, which limited their usefulness. The new sol-gel formulation is a major advance that offers a fast response rate of less than five seconds, very stable calibration (good for over a month with only a single baseline correction), and can easily be incorporated into a variety of product packages.
In addition, the sol gel can be applied to probes or as a coating to cuvettes, Petri dishes, microtiter plates, flow cells, or other media where the sample volume may be limited. Unlike other optical pH systems that measure fluorescence, this system is ratiometric and immune to drift; the sensors can be stabilized by steam or gamma radiation; and they can be used with O2 optrodes to provide pH and O2 sensing in a small common package. Furthermore, these optrodes can use "off-the-shelf detection"—miniature fiber-optic spectrometers, or existing spectrophotometers.
Applications include bioreactor monitoring (see Laser Focus World, March 2008, p. 65), cell-culture monitoring, waste-water discharge monitoring, atmospheric/acid rain measurement, medical-device applications for blood and body-fluid measurements, and field-portable applications, to name a few.
REFERENCES
1. W. Wang, C.E. Reimers, M.R. Shahriari and S.C. Wainright, The Marine Technology Society Annual Conference Proceedings 1 (1998).
2. M. Krihak, M. Murtagh and M.R. Shahriari, Chemical, Biochemical and Environmental Fiber Sensors VIII, Proc. SPIE 2836 (1996).
3 "Upendra H. Manyam (Ph.D. candidate), M.R. Shahriari (thesis advisor) and George Sigel (thesis co-advisor), Application of Dye-Doped Sol gel for pH Sensing, Ph.D. thesis, Rutgers University, NJ (March 2006).
About the Author
Bob Kearton
Bob Kearton is director of the OEM Business Unit at Ocean Optics (Orlando, FL).
Mahmoud Shahriari
Mahmoud Shahriari served as research director at Ocean Optics (Orlando, FL) from 1999 through 2012.

Jason Eichenholz
Jason Eichenholz is the cofounder and CEO of Relativity Networks and a pioneer of laser- and optics-enabled innovation, product development, and commercialization. He holds a Ph.D. in optical sciences and engineering, as well as more than 90 patents within the field. Eichenholz co-founded Luminar, a leading provider of LiDAR. In 2023, he co-founded Relativity Networks.