MEDICAL DIAGNOSTICS: Cell elasticity can predict metastasis
By comparing the difference in elasticity between malignant and nonmalignant cells, a new laser-based cancer test may be able to determine whether cancer has spread, and thus reduce the number of unnecessary mastectomies in women with breast cancer. Josef Käs, director of the Institute for Soft Matter Physics at the University of Leipzig, Germany, and Jochen Guck, a researcher there, have developed a shoebox-size laser-based “optical stretcher” that measures a cell’s elasticity, instead of its biological makeup, to determine whether the cell is cancerous.
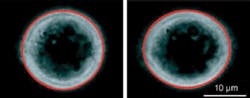
Cancer cells tend to dedifferentiate, losing the special characteristics of the organ in which they started life. Thus, they no longer need the rigid cytoskeleton that holds them in shape, making them more elastic than normal cells (see figure).
“A healthy cell is like a Gummi Bear, with a cytoskeleton like tent poles,” Käs said. “If it is cancerous, it loses the tent poles and becomes a fluid bag. And the softer the cancer cells, the more likely they are to travel through the body and produce secondary tumors (metastases).”
In the absence of a strong cytoskeleton, cancerous cells extend 40% more easily than their healthy counterparts, according to Käs. Cells from metastasized tumors are an additional 30% more elastic than early-stage cancer cells. Thus, the laser-based optical-stretcher method could potentially assess how far the disease has advanced and determine metastasis based on cell elasticity alone. While cancer-causing genes produce a small change in a cell’s structural proteins, the cell’s elasticity properties undergo dramatic changes, according to Käs. Given a sample that contains at least 50 tumor cells, the laser can spot cancer more than 90% of the time.
Fast measurement
The optical stretcher uses a beam of IR laser light (1060 or 800 nm) to test the physical strength of each cell, measure the progress of a cancer by examining only the primary tumor, and provide a diagnosis using as few as 50 cells, instead of the 10,000 to 100,000 required by other methods. With such small samples, diagnoses can be made even before solid tumors develop or where a traditional biopsy is problematic. Because it can test as many as 3600 cells per minute, the optical stretcher is fast enough to be useful in clinical diagnosis. The sample is measured in two hours and provides a distribution of all of the stretchiness of the cells in four hours. Käs started with a Ti:sapphire laser, but found a 1060-nm fiber laser more in line with cost considerations for a medical device.
As Käs explained, the cell is pretty much transparent, so light is not reflected or absorbed. When a laser beam from the device enters a cell, the light gains momentum because of the different conditions inside. According to the law of conservation of momentum, the cell must lose an equal but opposite amount, so its membrane bends towards the light source.
“The cell gets a kick backward,” Käs said. “When the laser light leaves the cell, it loses momentum and drags the cell’s opposite edge along with it. By applying a continuous beam, the cell gets stretched to its limit.”
The optical stretcher differs from optical tweezers, in which the light is focused to a sharp point to grab hold of a cell. Because the optical stretcher does not use focused light, laser beams strong enough to detect stretching can be used without killing the cell. The microfluidic chamber uses two fiber lasers or splits light from a single fiber laser in two. The configuration is a simple optical trap. The system can be set up on any cell-culture microscope using a CCD camera, a microfluidic pump system or syringe pump, a fiber laser, and a computer to control the procedure.
Other potential uses for the optical stretcher include diagnosing oral cancer and locating stem cells.