Microscopy: Multicontrast nonlinear imaging enables tumor microenvironment discovery
Although understanding of a tumor's microenvironment (the neighboring cells, molecules, and vasculature that support its growth) has had a major impact on cancer research, lack of an appropriate imaging method has hampered progress. Now, scientists at the University of Illinois Urbana-Champaign have announced multicontrast nonlinear imaging, an approach they have used to gain new insight into how malignancy starts and spreads.1
Besides allowing a view of a tissue's inherent nonlinear optical properties, the method facilitates simultaneous visualization of such early signs of development as angiogenesis, extracellular matrix reorganization, and non-native cell recruitment. The technique has shown that each of these events requires concurrent changes: enrichment of specific extracellular vesicles and a metabolic switch toward biosynthesis. This concurrence reveals detail about cancer development, and may enable a new strategy for diagnosis and treatment.
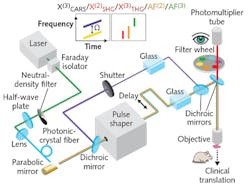
Pulse-shaping multiphoton microscope
To apply the technique, the researchers developed a pulse-shaping multiphoton microscope. The short-wavelength edge of the instrument's generated supercontinuum (780-880 nm), reflected by a dichroic mirror (DMLP900, Thorlabs), serves as the pump beam for coherent anti-Stokes Raman scattering (CARS). The main supercontinuum (900-1300 nm), directed to a commercial pulse shaper, is the CARS Stokes or excitation beam, which generates contrast from AF(2), AF(3), χ(2) SHG, or χ(3) THG (see figure). A computer-controlled line applies optical delay to the Stokes beam to enable spectral focusing CARS, and to collect the vibrational contrast of χ(3) CARS.
A pair of SF57 glass blocks, placed in the pump/Stokes beams, prechirps the pulses. Another dichroic mirror then recombines the two beams, steers them into a commercial microscope (BX61WI, Olympus), and focuses them using a super-apochromat objective (UPLSAPO 60× W/IR; numerical aperture, 1.20; Olympus). In AF(2)/AF(3)/χ(2) SHG/χ(3) THG imaging, the CARS pump beam was blocked by a computer-controlled shutter . A pixelated 4f pulse shaper (MIIPS Box640, Biophotonic Solutions) was used to iteratively compress the supercontinuum pulse, whereas the pulse measurement based on multiphoton intrapulse interference phase scans (MIIPS) was used in the iteration loop. The researchers validated the supercontinuum's coherence using converged iteration toward transform-limited pulse compression. They used local compression to generate tunable ~30 fs pulses across the supercontinuum's bandwidth and, following transform-limited pulse compression, programmed the pulses for customized multiphoton modalities.
The team then shaped these customized excitation pulses (using amplitude and phase), and applied various detection filters to target different endogenous molecules or structures of interest. They attenuated the incident power of the phase-shaped supercontinuum pulses by amplitude shaping to conservatively safe levels. They were able to repeatedly image individual specimens using different modalities without any observable sample damage.
They validated each of the multiphoton imaging modalities by performing power-dependence tests with unstained rat tissue (cancerous and normal). They acquired images (380 × 380 pixels, 0.5 × 0.5 μm2 per pixel) using different average powers, and calculated and plotted the average intensity (photon counts) against excitation average powers on double logarithmic scales. The linearly fitted slopes were consistent with the quadratic power dependence for AF(2) and χ(2) SHG, the cubic power dependence for AF(3) and χ(3) THG, and the quadratic/linear power dependence in pump/Stokes for χ(3) CARS. The researchers suggest that a few factors may account for the <10% deviation from theoretical numbers: the specimens' diverse biomolecular composition, excitation/emission cross-talk, and low or saturated signal in the field of view.
The system acquired images by raster scanning a piezoelectric stage-for all modalities, pixel dwell time was 200 μs. To generate a strong, high-spectral-resolution χ(3) CARS signal, they used spectral focusing to concentrate the supercontinuum's spectrally dispersive power into a single vibrational frequency. To coherently drive one molecular vibration, the system chirps both the pump and Stokes pulses so that their instantaneous frequency difference remains constant during interaction time.
Scanning the delay between two pulses to tune the frequency difference with a spectral resolution of 14 cm-1 enabled spectroscopic signal acquisition. The system collects hyperspectral images—at a single Raman vibration frequency that varies by image—using spectral-focusing CARS. Then, it calculates the χ(3)CARS spectrum from these images over pixels corresponding to the region of interest.
The method promises to provide insight on the complex interrelations and possible causal links involved in carcinogenesis and metastasis.
REFERENCE
1. H. Tu et al., Sci. Adv., 3, 1, e1600675 (2017); doi:10.1126/sciadv.1600675.
About the Author

Barbara Gefvert
Editor-in-Chief, BioOptics World (2008-2020)
Barbara G. Gefvert has been a science and technology editor and writer since 1987, and served as editor in chief on multiple publications, including Sensors magazine for nearly a decade.