IR Spectroscopy/Cancer Theranostics: Spectroscopy developments yield molecular pathology
The past two decades have seen vibrational spectroscopy advance to the point of enabling a new diagnostic approach. Called molecular pathology, the method analyzes a whole specimen—including its entire genome, proteome, and metabolome—rather than simply probing tissue for the presence of selected biomarkers, as standard pathology techniques do.
Conventional histopathology involves visual inspection of stained, biopsied specimens that are either formalin-fixed and paraffin-embedded, or else unfixed and flash-frozen. It uses the pathologist's eyes as the detector and relies on "databases" stored in his or her memory to discriminate and diagnose.
Molecular pathology, meanwhile, requires minimal sample preparation and visualizes the "spectral vector": a map of infrared (IR) absorption intensities vs. absorbed radiation wavelength for all the biochemical components comprising each subcellular-sized pixel sampled. The approach is based on the fact that transition from healthy tissue to diseased states corresponds not only to the morphology and tissue architecture changes observed by standard histopathology, but also to a shift in the tissue's overall biochemical composition—which is revealed by changes in IR spectra.
Improved cancer diagnosis, treatment
Two variants of molecular pathology are spectral cytopathology (SCP), which focuses on cells, and spectral histopathology (SHP), whose target is tissue. Recent studies led by Max Diem at Northeastern University (Boston, MA) and Cireca (Cambridge, MA) have demonstrated the efficacy of IR SHP for discriminating between benign and cancerous tissue in lung tumors.
A pilot study, which examined 80 patient samples in tissue microarray (TMA) format, showed SHP's ability to classify the most common types of lung cancer: small-cell lung carcinoma (SCLC), squamous-cell carcinomas (SqCCs), and adenocarcinomas (ADCs).1
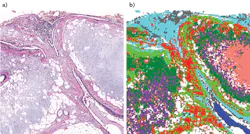
A 2015 publication reports follow-up research involving 449 samples, including tissue biopsied from cancer patients, plus benign lung-tumor samples and lung specimens determined to be cancer-free (see figure).2 This study covered the original differentiation of the three cancer types, and also evaluated the ability to identify necrotic tissue from the three cancers and to classify ADC subtypes, SqCC grades, and four classes of nonmalignant lesions. The publication describes acquisition of IR datasets including thousands of spectra, each collected from tissue pixels approximately 6 μm on edge.
Together, the pixel spectra produce a comprehensive reading of the biochemical composition of the entire sampled area. Subsequent multivariate analysis of the hyperspectral datasets revealed biochemical changes not only among tissue types, but also among various stages and states of disease. The results suggest that SHP's diagnostic accuracy is comparable to that of multipanel immunohistochemistry.
Among SHP's major advantages is the fact that it can easily integrate into the existing pathology workflow. In addition, the technique is label-free and nondestructive, requires minimal sample preparation, and offers traceability along with high spatial resolution. The method offers reproducibility, complete objectivity, and improved accuracy. Thus, it represents a major innovation in medical diagnostics.
Going clinical
Now, Cireca is pursuing commercialization of SHP using instrumentation based on commercial imaging IR spectrometers. While Cireca's scalable platform can quantify and compare many tissue types, the company is focusing its initial application on the focus of the research described: advanced identification of lung cancer types. From every 2-mm-diameter tissue spot, the automated system collects 100,000 pixel spectra, each of which includes 450 intensity values.3
Cireca has developed novel methods and algorithms to extract high-content diagnostic, prognostic, and predictive (therapeutic) information that promises pathologists more accurate insights on disease pathology and the ability to develop more personalized, effective treatments than are currently possible. The goal is to significantly enhance cancer diagnosis, staging, prognosis, therapy selection, and monitoring within existing pathology workflows.
Cireca is currently seeking collaboration with research centers and institutions to further advance its technology, and is also exploring applications in other industries.
REFERENCES
1. B. Bird et al., Lab. Invest., 92, 1358–1373 (2012).
2. A. Akalin et al., Lab. Invest., 95, 406–421 (2015).
3. X. Mu et al., Analyst, 140, 2449–2464 (2015).
About the Author

Barbara Gefvert
Editor-in-Chief, BioOptics World (2008-2020)
Barbara G. Gefvert has been a science and technology editor and writer since 1987, and served as editor in chief on multiple publications, including Sensors magazine for nearly a decade.