MULTISPECTRAL DETECTORS: Multispectral system evaluates treatments in dermatology
An advanced multispectral dermatascope can be used to study changes in the macroscopic structure of the skin. The system produces detailed images that can be used to track skin changes over time.
ROWLAND de ROODE, HERKE JAN NOORDMANS, RUDOLF M. VERDAASDONK, AND VIGFUS SIGURDSSON
Optical diagnosis and therapy are becoming effective methods for evaluation and treatment of a broad variety of medical conditions. The University Medical Center, Utrecht (Utrecht, The Netherlands), has developed a spectral imaging device to perform quantitative per-pixel spectral analysis of tissue for evaluation of laser treatment of skin lesions, or for tracking skin diseases as they heal over time. The current systems, called dermatoscopes, are based on standard red/green/blue (RGB) charge-coupled-device (CCD) detectors. However, because skin has more useful spectral properties in the visible spectral range-like the absorption peaks of hemoglobin near 540 and 580 nm-a multispectral dermatoscope can provide improved functionality.
Multispectral dermatoscope
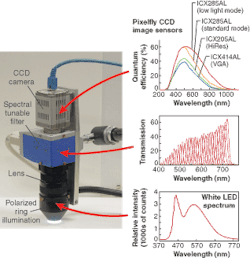
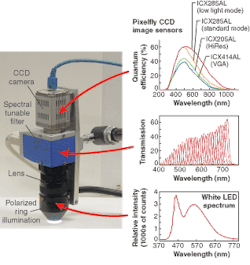
A 5-W Luxeon white LED (Lumileds; San Jose, CA) source is fiber-coupled to a ring-shaped polarized illuminator mounted on the camera head of the multispectral dermatoscope. A light shield excludes any other light and maintains a standard focal distance. Cross-polarized illumination is used to exclude direct reflection of skin tissue.
By inserting a liquid-crystal tunable filter (LCTF) between lens and camera, a user can capture a spectrally filtered image at any given wavelength between 400 and 720 nm with a bandwidth of approximately 10 nm(see Fig. 1).1 The LCTF is based on a Lyot filter and therefore has a polarizing effect. Its polarizing angle is crossed with respect to the orientation of the illumination polarization so that direct surface reflections are eliminated and the camera only sees scattered light.2 The spectrally filtered images are captured by a compact, high-resolution, 12-bit monochrome CCD camera.
Although the multispectral camera has a spectral range between 400 and 720 nm, the LED illuminator only gives enough light for spectral imaging in the range between 440 and 720 nm. Because the bandwidth of the LCTF is approximately 10 nm, a step size of 3 nm is chosen as a standard.
System calibration and optimization
The multispectral system must be carefully calibrated to obtain reproducible measurements. When a constant exposure time is used, the sensitivity spectrum of the system is far from flat. Because of the strong nonuniform character of the sensitivity spectrum, the signal-to-noise ratio (S/N) will be far worse for ultraviolet (UV) than for green wavelengths. To achieve uniform S/N across the spectrum, the acquisition time in the UV region must be longer than in the green region. The best acquisition time per wavelength is sequentially determined using a barium sulphate white reference object. This time is determined by applying a loop that automatically alters integration time per wavelength and achieves the best S/N across the spectrum.
The spectrum of the acquired image data is determined by the illumination spectrum of the LED, the reflective spectrum of the skin, the transmission spectrum of the LCTF, and the sensitivity spectrum of the CCD. As only the reflection spectrum is of interest and not the spectra of the system components, a full spectral scan of the barium sulphate white reference is again made to divide out these unwanted sensitivity spectra. The relative reflection is then given by:
Here, the dark reference value corresponds to the signal offset of the CCD. By using the white reference correction for every pixel individually, small spatial inhomogeneities in the illumination distribution are corrected in the same step.
Treatment evaluation
For clinical experiments, the multispectral system is placed on a compact trolley with an isolation transformer for portable use. The camera is mounted on a mechanical arm to enable fast and fixed positioning of the camera head on the skin of the patient.
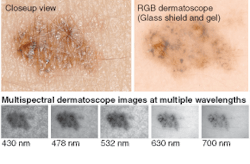
To evaluate the effectiveness of the multispectral imaging system, a study at the Outpatient Department of Dermatology at University Medical Center, Utrecht, compared the performance of a normal color dermatoscope and the multispectral dermatoscope on a port wine stain (see Fig. 2). Moving from UV to IR wavelengths in a multispectral image, light is scattered deeper in the tissue near the UV range, while deeper skin structures are observed near the IR wavelengths. At the same time, the reflection spectrum is modulated by the absorbing fluorophores in the skin like melanin and hemoglobin.
Port-wine stains appear red due to hypervascularization-an overabundance of small blood vessels in the skin. The laser treatment for this condition consists of destroying the micro vessels by taking advantage of the spectral absorption characteristics of hemoglobin running through the blood vessels.3 A typical laser treatment consists of high-energy pulses (532 nm, 12 J/cm2, 30-ms pulse width) with a spot size of approximately 5 mm. The laser light is delivered by a handpiece with a cooled glass shield that is placed onto the skin with transparent gel in between for better optical coupling of the green laser light. Because of the high spectral absorption of the microvessels at 532 nm, only microvessels are destroyed and surrounding tissue remains intact. These laser treatments usually need to be performed many times with an interval of two months between treatments.
Special software can be used to relocate the same area of interest and acquire multispectral dermatoscopic images reproducibly during these long-term laser treatments. The decrease of vascularization over time of distinct blood vessels can be visualized and quantified.
Clinical diagnosis
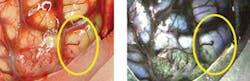
The next step in the technical development of the multispectral dermatoscope is enhanced visualization. A false-color image, for instance, that indicates the level of suspected malignant tissue can provide improved diagnosis before biopsy or treatment of invasive tumors. As an example, malignant brain tumors on a human cortex were imaged during surgery. A false-color visualization was created by putting three spectrally filtered images into the red, green, and blue color components of a color image. The resulting image showed much better tumor demarcation than the standard RGB CCD image (see Fig. 3). A three-chip CCD camera coated with these particular spectral filters could be a useful tool for the surgeon removing the tumor.
Image motion is a major problem when acquiring multispectral data in vivo. Motion can occur when a patient moves during acquisition or when two multispectral data sets acquired at different moments in time are compared. Motion correction can be done manually or automatically, but is not always easy because of the complexity of the vasculature.4 In addition, the match must have elastic properties to accommodate the flexible properties of human skin, which again makes the matching problem more difficult. This type of image processing demands much of the computer-the match of a full-resolution multispectral dataset can take up to a full day. One possible solution for this might be to use three-dimensional (3-D) graphics cards.5 The graphics processing unit (GPU), originally developed for 3-D games, can perform elastic matches much faster.
Multispectral data is also more difficult to interpret because humans are not accustomed to perceiving more than three color bands at the same time. To optimally use all information, application-dependent image processing must be performed using a combination of spatial pattern recognition and spectral analysis.
Despite some technical difficulties, the Utrecht group has successfully acquired multispectral data for in vivo image analysis, and is currently optimizing solutions to correct for image motion during acquisition. These advances are further enabling clinicians to use multispectral imaging for the evaluation of dermatological treatments and disease diagnosis over time.❏
REFERENCES
1. R. de Roode, H.J. Noordmans, and R.M. Verdaasdonk, Proc. SPIE6078, A-28 (2006).
2. C. Balas, IEEE Trans Biomed Eng.48(1) 96 (2001).
3. C. Clark et al., Lasers in Medical Science19, 1 (2004).
4. H.J. Noordmans, R. de Roode, and R.M. Verdaasdonk, Proc. SPIE6081, 6 (2006).
5. R. Strzodka, M. Droske, and M. Rumpf, J. Medical Informatics and Technologies6, 43 (2003).
ROWLAND de ROODE, HERKE JAN NOORDMANS, and RUDOLF M. VERDAASDONK are researchers in the Department of Medical Physics, and VIGFUS SIGURDSSON is a researcher in the Department of Dermatology, all at University Medical Center, Utrecht, Room F.01.125, P.O. Box 85500, 3508 GA Utrecht, The Netherlands; e-mail: [email protected].