OPTOGENETICS/NEUROSCIENCE: Light triggering demonstrates that memory engrams are physical phenomena
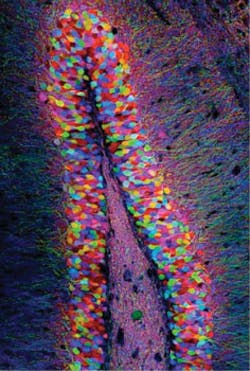
Memories reside in specific brain cells. That's the finding of Massachusetts Institute of Technology (MIT; Cambridge, MA) researchers, based on its use of optogenetics—a technique that uses low-level light to selectively activate proteins administered to live tissues. The scientists showed that behavior based on high-level cognition can be generated by activating a few brain cells using light, says Susumu Tonegawa, lead author of a paper describing the study.1
The researchers first identified a specific set of brain cells in the hippocampus of mice that were active only when a mouse was learning about a new environment. They determined which genes were activated in those cells, and coupled them with the gene for channelrhodopsin-2 (ChR2), a light-activated protein. Next, they studied mice with this genetic couplet in the cells of the dentate gyrus of the hippocampus, using tiny optical fibers to deliver pulses of light to the neurons. The light-activated protein would be expressed only in the neurons involved in experiential learning—allowing for labeling of the physical network of neurons associated with a memory engram for a specific experience.
They then put the mice into a new environment and allowed a few minutes of exploration before administering a mild foot shock—which led the mice to fear the environment. The brain cells activated during this fear conditioning became tagged with ChR2. Later, when exposed to triggering pulses of light in a completely different environment, the neurons involved in the fear memory switched on—and the mice adopted a defensive crouch.
The method may have applications in the study of neurodegenerative and neuropsychiatric disorders.
1. X. Liu et al., Nature, 484, 381–385 (2012).
More BioOptics World Current Issue Articles
More BioOptics World Archives Issue Articles