Three-photon microscopy comes of age
Two-photon microscopy (2PM) was first reported in 1990 using 100 fs pulses from a dye laser in lieu of single-photon excitation with a continuous-wave laser.1 Two inherent advantages were immediately apparent: the longer excitation wavelength used in 2PM decreased photobleaching and photodamage, while the nonlinearity of the process provided optical sectioning without requiring a confocal pinhole.
It soon became evident that the lower attenuation of infrared light within biological tissue enabled deeper imaging as well. The high transparency of brain tissue made 2PM an essential tool for functional brain research in model animals like C. elegans worms, fruit flies, zebrafish, and mice. Other optical techniques like optical coherence tomography and photoacoustic imaging can image even deeper, but cannot achieve the cell-level resolution of 2PM.
Most dyes for confocal imaging were developed to match the ultraviolet, blue, and green lines of ion lasers, prevalent in the 1980s and 1990s. But 2P excitation requires photons with half the energy, at near-infrared wavelengths ranging from 700 to 1000 nm. The widespread adoption of tunable titanium sapphire (Ti:sapphire) mode-locked lasers shortly after 1990 proved to be a good match for 2PM: the ~100 fs pulse duration provides efficient fluorophore excitation, while the typical 80 MHz pulse repetition rate enables fast scanning. Furthermore, the 300 nm wavelength tuning range covers most fluorescent probes.
In the pursuit of ultimate imaging depth, in 2003, a team led by Winfred Denk imaged living mice brains to a depth of 1 mm. They accomplished this feat by replacing the Ti:sapphire laser with a regenerative amplifier running at 200 kHz with pulse energy in the microjoule range, about 100 times higher than the Ti:sapphire oscillator.2 However, the complexities of regenerative amplifiers prevented this approach from being adopted.
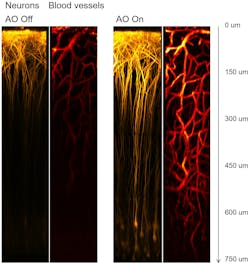
The use of high-energy, low-repetition rate excitation for deep imaging remained unexploited until 2013 when Chris Xu’s team at Cornell realized that three-photon microscopy (3PM) leverages short, energetic pulses to a larger extent than 2PM.3 Since then, 3PM gained popularity as a tool able to extend the capabilities of 2PM by imaging deeper layers in the brain and other tissues such as tumors and bone.4, 5 In an example of 2P vs. 3P images with increasing depth (see Fig. 1), the contrast difference becomes striking past 550 µm. Furthermore, 3PM can even penetrate the skull (see Fig. 2).
We asked Dr. Jack Waters, associate director of the Imaging Department of the Allen Institute for Brain Sciences, for his opinion on the intricacies of 3P imaging. He says 3PM is now at the same stage where 2PM was 5 to 7 years after its invention when users were still struggling to get useful images.
Depth advantage of 3P
Imaging depth in 2PM is limited by scattering and absorption of the excitation light within the tissue. As the imaging plane is positioned deeper, fewer photons reach the focal volume, but the loss of light can be compensated by increasing the laser power. However, the power at the sample surface and the top layers may eventually cause damage. Even if the power is kept below the damage threshold, as the imaging plane gets deeper, the excitation beam will generate a low-efficiency background in the conical volume from the sample surface to the imaging plane. At a certain depth, this background overcomes the signal and sets a limit to the imaging depth.
Once this signal-to-background limit is reached, increasing the excitation power will no longer improve the image quality. 3PM overcomes this limit because the higher nonlinearity of the 3P excitation reduces the background. Moving from 2PM to 3PM requires changes in the microscopy setup, as we describe in the following.
Excitation wavelength selection
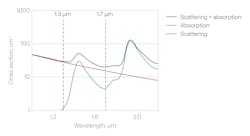
3P excitation requires longer wavelengths than conventional 2PM because the same energy transition is achieved with three rather than two photons. For example, ubiquitous green fluorescent protein and brain activity indicators (GCaMPs) are excited at 920 to 940 nm in 2PM, but require 1300 nm in 3PM. Red-shifted probes excited at ~1100 nm in 2PM benefit from 3P excitation at 1700 nm. Fortunately, 1300 nm and 1700 nm are at the local minima of light attenuation (combination of absorption and scattering) in water (see Fig. 3).
While scattering decreases monotonically at longer wavelengths, absorption depends on the chemistry of the sample, usually dominated by water. Water absorption increases going from 920 nm to 1300 nm and 1700 nm, but the decrease in scattering gives an advantage to these longer wavelengths. Absorption, however, limits the permissible power because it leads to tissue heating and ultimately damage.
As Waters explains, at 920 nm, it is reasonably safe to put 250 mW of laser power into a mouse brain, but it should not exceed 100 to 150 mW at 1300 nm, and half that value at 1700 nm. In his experience, 1300 nm is more widely used in brain imaging because red-shifted functional probes yield lower signal than GCaMP. Some probes can even be excited at 1300 nm, and objective performance is generally better at this wavelength. For intravital imaging, 1700 nm offers benefits such as simultaneous 3P and 4P excitation of fluorophores from blue to the far-red and third-harmonic generation (THG) imaging inside the bone.4, 5
Tunable sources continue to dominate 2P imaging applications, although there is an increasing uptake of fixed-wavelength lasers. It is too soon to determine what wavelength tunability is required for 3P imaging. However, even for applications concerned only with 1300 nm, a central wavelength tuning range of 50 to 100 nm proves beneficial. With improvements in the brightness of red-shifted probes, the use of 1700 nm light may allow imaging even deeper with single-cell resolution. While fixed-wavelength lasers for 2PM may be more stable, compact, and cost-effective, this tenet does not apply to 3P imaging because laser sources at 1300 nm are different, and different technical tradeoffs apply.
Pulse duration and compression
Lasers for 2PM produce pulses with 100 to 150 fs duration. 3P imaging requires pulses of about 50 fs because the higher nonlinearity of the 3P process depends critically on the peak power of the pulses. In most cases, longer pulses simply will not provide sufficient depth advantage to make 3P worthwhile compared to 2P.
Unfortunately, the generation of such short pulses is only half of the challenge. Short pulses have a large spectral bandwidth and experience group delay dispersion (GDD) when they propagate through the material, which causes the different spectral components to spread out in time. This reduces the peak intensity and the multiphoton signal strength, even though the average power stays the same.
To produce optimal signal, the GDD in the optics from laser to sample needs to be pre-compensated (pre-chirped) with a pulse compressor. Halving the pulse duration doubles the bandwidth and makes the design of compressors more challenging. In fact, Waters explains that as 3PM gained momentum, the main difficulty encountered by users is managing the pulses—from building a suitable compressor (which initially was not provided by laser manufacturers) to ensuring that the pulses have the shortest duration at the focal plane. Inadequate pulse compression is often the difference between collecting a signal and burning the sample with no signal at all.
Still, Waters says there is room to optimize laser parameters. For example, little work has been done to explore whether pulses shorter than 50 fs are beneficial, although managing the dispersion of even shorter pulses may be challenging.
Pulse energy, repetition rate, and average power
Differences in excitation cross-section between 2PM and 3PM require more than just shortening the pulses to 50 fs. They also require a substantial increase in energy per pulse. Beyond this, microscope objectives and other elements are not yet fully optimized for high transmission and low dispersion at 3P excitation wavelengths. It translates into higher losses in the optical train—as much as 90%. These and previous considerations highlight that we must carefully balance peak power (pulse energy), average power, and repetition rate. We know that 100 mW is a good target for damage-free sample illumination.
With 10% microscope throughput, the laser should produce about 1 W of power. Should this power be administered as 1 μJ pulses at 1 MHz or rather as 100 nJ at 10 MHz? So far, most 3P imaging is performed at 1 MHz with a dwell time of several microseconds and one or several laser shots per pixel to provide sufficient signal at a frame rate of a few hertz.
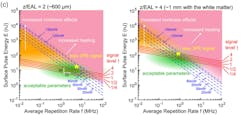
Resonant scanning would benefit from higher repetition rates, but the increase in average power simply via a higher repetition rate may damage the sample. Waters believes careful optimization could support resonant scanning at 4 to 5 MHz. And, the optimum repetition rate as a function of tissue scattering and imaging depth is discussed extensively by Wang and Xu (see Fig. 4).6
A high pulse energy results in a bright multiphoton signal that can be observed even in highly scattering tissues at depth, but energies too high can result in nonlinear damage. A high average power and high repetition rate also increases the signal and enables high-speed imaging, but excessive power results in too much heating. These factors leave a rather narrow window for 3P imaging which varies with sample and imaging depth, complicating the choice of parameters for a 3P laser.
In Figure 4, you can see how the optimal parameter range (shown in green) changes with increasing depth—a higher repetition rate is better at 600-µm depth, and 1 MHz is better at 1 mm. While 2PM uses lasers at 80-MHz repetition rates, the lower repetition rate and higher energy of a 3PM laser require a different approach. This involves a more energetic laser amplifier, usually operating at 1030 nm, and pumping a tunable device called an optical parametric amplifier (OPA).
Although wavelength-tunable, OPAs generally operate at a fixed pump energy. The repetition rate and output power of the OPA can be increased or decreased, but the energy must stay the same and provide sufficient margin at the desired deepest imaging point. Imaging shallower planes is then accomplished by reducing the power with an external modulator. The deepest layer sets the required maximum energy, while the fastest desired imaging speed sets the maximum required repetition rate—two specifications that are difficult to fulfill in a single device.
Until recently, lasers for 3PM were a combination of separate boxes housing an ultrafast amplified pump laser, an OPA, and an external, frequently home-built compressor usually provided by different manufacturers and assembled on the optical table, which often came with compromises in stability, maintenance, and specification assurance.
It’s evolved with the introduction of integrated systems such as the CRONUS-3P by Light Conversion. In these microscopy sources, every component is manufactured and tested under the same roof, streamlining much of the laser setup process for 3PM. It brings the setup closer to the single-box 2P sources to ensure seamless functionality.
Energy and long-term average power stability, along with beam mode quality, are also crucial factors. In the one-shot per pixel regime for fast 3P imaging, shot-to-shot laser noise becomes especially important because it gets imprinted in the image.7 Adaptive optics have demonstrated great potential for enhancing imaging depth and contrast due to the high nonlinearity of 3P excitation.8
Road ahead
The advent of single-box and turnkey tunable lasers helped popularize 2P microscopy by eliminating the need for a laser expert in imaging laboratories. A similar trajectory is anticipated for 3PM, with 3P laser sources getting more reliable, tailored to microscopy user requirements, compact, and less complex and costly to operate.
Even with a reliable laser, setting up a 3P microscope is more involved than a 2P rig, and it is important to also have a 2P setup in the same lab for debugging and validating the samples. With a tunable 3P source, the excitation wavelength and the imaging parameters can be further optimized. An interesting alternative is modifying the pulse spectrum directly via self-phase modulation through controlled bending of a fiber as demonstrated by Sixian You’s group at MIT.9
In terms of imaging speed, Waters points out notable developments such as fast volumetric imaging by Alipasha Vaziri at the Rockefeller Institute, and Chris Xu’s adaptive excitation source (AES) where the laser operates not at a steady repetition rate but producing pulses only when the scanner is over a neuron.10, 11 This enables a higher instantaneous repetition rate while adhering to the average power limit, but requires a complex feedback loop from the sample to the laser. While these advancements are currently at the proof-of-principle stage, the imaging community, including Waters, looks forward to their future adoption in neuroscience labs. He concludes that his work is also in the direction of fast switching of a 3P laser source to control the average power deposition in the sample while maintaining the imaging speed.
REFERENCES
1. W. Denk, J. H. Strickler, and W. W. Webb, Science, 248, 4951 (1990); https://doi.org/10.1126/science.2321027.
2. P. Theer, M. T. Hasan, and W. Denk, Opt. Lett., 28, 12 (2003); https://doi.org/10.1364/ol.28.001022.
3. N. G. Horton et al., Nat. Photonics, 7 (2013); https://doi.org/10.1038/nphoton.2012.336.
4. G.-J. Bakker et al., eLife, 11, e63776 (2022); doi:10.7554/elife.63776.
5. D. Tokarz et al., PLoS ONE, 12, 10 (2017); doi:10.1371/journal.pone.0186846.
6. T. Wang and C. Xu, Optica, 7, 8 (2020); https://doi.org/10.1364/optica.395825.
7. L. Kontenis et al., Proc. SPIE, PC12384, PC1238410 (2023); https://doi.org/10.1117/12.2649932.
8. C. Rodríguez et al., Nat. Methods, 18, 10 (2021); doi:10.1038/s41592-021-01279-0
9. T. Qiu et al., Nat Commun., 15, 1 (2024); doi:10.1038/s41467-024-46244-5.
10. S. Weisenburger et al., Cell, 177, 4 (2019); doi:10.1016/j.cell.2019.03.011.
11. B. Li et al., Nat. Methods, 2 (2019); doi:10.1038/s41592-019-0663-9.
12. O. A. Shemesh et al., Nat. Neurosci., 20, 12 (2017); doi:10.1038/s41593-017-0018-8.
13. V. Emiliani et al., J. Neurosci., 35, 41 (2015); doi:10.1523/jneurosci.2916-15.2015.
High-energy pulses for holographic photostimulation
Lasers producing high-energy pulses at a low repetition rate have found applications elsewhere in microscopy; for example, in all-optical multiphoton control and readout of neural circuits.12, 13
To stimulate neurons, excitation light needs to be sculpted with single-neuron spatial resolution, which is achieved using widefield holographic techniques and millisecond temporal resolution, which is straightforward for amplified lasers with integrated pulse-pickers.
Holographic stimulation is lossy and requires multiple watts of average power to stimulate large groups of neurons simultaneously. Fortunately, many opsins can be excited by two photons at 1030 nm, which current lasers can provide with powers exceeding 100 W, and these are the same lasers that pump OPAs used for three-photon imaging. Therefore, it is possible to use the same laser for stimulation at 1030 nm and 3P readout at 1300 nm or to tune the OPA to optimize opsin absorption; e.g., at 960 nm.
About the Author
Lukas Kontenis
Lukas Kontenis is an R&D Engineer at Light Conversion (Vilnius, Lithuania).
Mantvydas Mikulis
Mantvydas Mikulis is a product manager at Light Conversion (Vilnius, Lithuania).
Gabrielė Stankūnaitė
Gabrielė Stankūnaitė is communications project manager at Light Conversion (Vilnius, Lithuania).
Jonas Berzinš
Jonas Berzinš is market development manager at Light Conversion (Vilnius, Lithuania).
Marco Arrigoni
Marco Arrigoni is vice president of marketing at Light Conversion (Vilnius, Lithuania). He previously served as director of marketing at Coherent (Santa Clara, CA) from 2007 through 2023.