Next-level virus detection via WIDE-MIP microscopy
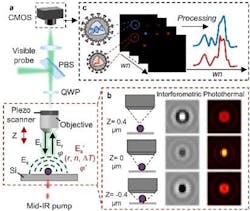
A team of Boston University (BU; Boston, MA) researchers developed a widefield interferometric defocus-enhanced mid-infrared (mid-IR) photothermal (WIDE-MIP) microscope (see video and Fig. 1) that performs fingerprint analysis of bio-nanoparticles. An associated theoretical framework the researchers created calculates the defocused interferometric photothermal images of single nanoparticles with different sizes—and provides the optimal mid-IR photothermal detection focus position relative to the nominal focus position of the particles.
Compared to conventional scanning methods, the researchers’ system has demonstrated vibrational detection of 100 nm polymethyl methacrylate particles at a signal-to-noise ratio level similar to conventional scanning methods—but with three orders of magnitude higher throughput.
“By simply tuning the infrared wavenumber, the WIDE-MIP spectra of single viruses are acquired from the hyperspectral images,” says Ji-Xin Cheng, the BU Moustakas Chair Professor in Photonics and Optoelectronics, who led the research.
Because photothermal signals are a modulation of visible beam intensity, it’s widely believed that maximized bright light contrast generates an ideal mid-IR photothermal contrast. It’s not the case with interferometric mid-IR photothermal microscopy, Cheng explains, in which a signal strongly depends on the relative phase between the particle-scattered photons and the substrate-reflected reference field.
“Instead, we’ve found that by fine-tuning the focus position of the objective, the defocused interferometric imaging results in a greatly improved MIP contrast,” Cheng says.
Facing challenges head-on
There is an “urgent need to develop tools for rapid and accurate virus detection,” Cheng says, citing the motivation behind his team’s years-long research project. “We’ve suffered from all kinds of viral pandemics over a long history—like flu, SARS, yellow fever, and so on. And the emergence of the monkeypox outbreak in early 2022 posed a new global health threat during the COVID-19 pandemic.”
Currently used diagnostic tests on viruses commonly rely on the detection of nucleic acids or surface proteins. However, this challenge has become more daunting because virus particles are extremely small and contain “a low abundance of chemical components. For example, there about 5000 proteins in a single vesicular stomatitis virus,” Cheng says.
The amount of viral nucleic acid in a single virion is generally lower than the amount of viral protein. Detecting viral nucleic acids is tough without signal amplification techniques, including polymerase chain reaction (PCR).
“Although nucleic acid amplification tests and antigen rapid diagnostic tests (RDT) can provide accurate testing results, they usually require pre-treatments of a large number of virions, extraction, or tagging that add time to any assay,” Cheng explains. “It is noteworthy that residual viral RNA from patient specimens remains detectable even when patients have recovered or have no culturable viruses. Thus, PCR or RDT may work great at the early stage but are a coin flip at the end.”
Conventional optical detection methods have shown potential clinical diagnostic value. Interferometric microscopy is among those used for single-virus tracking, viral infection study, and demonstration of translation into clinical settings. But these methods lack molecular information of the viruses, while the chemical contents are critical to viral structure and function. This makes new complementary methods crucial to identify the intact virions with preserved structure to confirm viral infection and reduce false diagnoses.
Several years ago, Cheng’s team developed a widefield MIP microscope for ultrafast chemical imaging. But it has limitations in detection sensitivity. “Existing widefield MIP microscopes could not meet the requirements for detecting single viral nanoparticles and performing precise spectral analysis,” he says.
The BU researchers have made some notable, successful improvements in the years since.
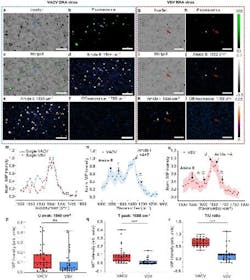
Their enhanced WIDE-MIP microscope touts a new key feature: advanced composition detection—essentially, the ability to identify the composition of viral nucleic acids and proteins in single viruses. The new microscope identifies absorption peaks in samples, revealing the specific contents of viral proteins and nucleic acids within DNA and RNA viruses. It can also reveal critical details about the structural components and chemical composition of viruses.
“WIDE-MIP enables the differentiation between DNA and RNA viruses,” Cheng says. “This provides a new avenue for understanding viral vectors and screening for virus-based diseases.”
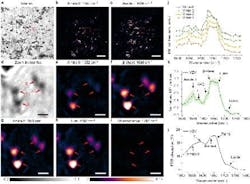
Insight into how proteins function is another plus with WIDE-MIP imaging, according to the researchers (see Fig. 2). It reveals critical details about the structural components of viruses, such as the enriched presence of the β-sheet—beta strands (β-strands) connected laterally by several backbone hydrogen bonds to form a generally twisted, pleated sheet beta sheet—components in DNA varicella-zoster virus proteins (which causes chickenpox and shingles).
“This information could be instrumental in elucidating the function of these proteins in virus assembly and virion activity,” Cheng says.
The WIDE-MIP technology allows high-throughput analysis. The researchers discovered an “enormous increase” in throughput compared to existing methods, Cheng says, citing scanning MIP systems. The WIDE-MIP microscope’s bandwidth is three orders of magnitude higher, making it a powerful tool for both clinical and research applications in the study of viruses (see Fig. 3).
Key to readiness
While the WIDE-MIP system has advanced significantly since its inception, the BU team has more work to do before it can be used in real-world settings.
The WIDE-MIP microscope for virus detection and analysis is currently still in the research and development phase. The team is exploring avenues for commercializing it, Cheng says, which includes further validation of the technology's reliability and robustness, scaling up production and distribution, and adapting it for practical clinical and diagnostic use.
“Our technology shows immense promise and has already demonstrated significant advancements in virus analysis,” Cheng says. “But it may take some time before it becomes a real product.”
The researchers are now addressing concerns about complex clinical samples, microfluidics, and antibody modified substrates for use in immobilizing specific viruses in complex media such as serum. For translation applications, a larger field of view is preferred when dealing with the clinical samples. In the current configuration of WIDE-MIP, Cheng explains that the IR pump beam is loosely focused, providing the effective imaging field of view (~50 µm) due to limitations in IR power density.
The team found that addressing such limitations means a more powerful IR laser with high energy and IR focusing optics with longer focal length can be used to increase the field of view.
“Our future improvements to WIDE-MIP will also focus on fingerprinting viruses or exosomes in liquid conditions, allowing for detecting biological nanoparticle samples in their natural states,” Cheng says, adding the researchers can achieve that by incorporating microfluidic systems and designed substrates to capture viruses and enhance imaging contrast in liquid measurements. “That will broaden the applicability of WIDE-MIP to real-world applications.”
FURTHER READING
Q. Xia et al., Nat. Commun., 14, 6655 (2023); https://doi.org/10.1038/s41467-023-42439-4.
About the Author
Justine Murphy
Multimedia Director, Digital Infrastructure
Justine Murphy is the multimedia director for Endeavor Business Media's Digital Infrastructure Group. She is a multiple award-winning writer and editor with more 20 years of experience in newspaper publishing as well as public relations, marketing, and communications. For nearly 10 years, she has covered all facets of the optics and photonics industry as an editor, writer, web news anchor, and podcast host for an internationally reaching magazine publishing company. Her work has earned accolades from the New England Press Association as well as the SIIA/Jesse H. Neal Awards. She received a B.A. from the Massachusetts College of Liberal Arts.