Using near-infrared spectroscopy to help unravel the mysteries of dementia
Researchers at the University of Cambridge, in conjunction with teams from University College London and the University of Sheffield (all in the U.K.), are using near-infrared (NIR) light—in the 600–1000 nm range—to look at different parts of the brain, such as the frontal lobe, where the working memory resides (see video). Light in this range travels relatively well through tissue and even better through bone, according to Gemma Bale, an assistant professor at the University of Cambridge and head of the Neuro Optics Lab.
With the NIR spectroscopy technique, the team also looks at oxygenated blood, which flows more in the active areas of the brain.
In particular, the team views hemoglobin, the protein in red blood cells that carries oxygen to the tissues. Hemoglobin, the best absorber of NIR light, absorbs light differently when bound to oxygen. Oxygenated blood is bright red, while deoxygenated blood is more of a purple color. When researchers shine the light into the brain, it scatters, with some bouncing back to the surface, allowing the researchers to detect the color.
“We're basically using this spectroscopy method in the brain to see where there’s oxygenated blood and use it as a metric of brain function,” Bale explains.
When the brain is more active, it recruits more energy, meaning more oxygenated blood will flow to those areas; there is little activity with dementia. So, by developing a technique that measures this flow, the researchers are establishing another quantifiable metric of brain function linked to dementia.
Exploring Alzheimer’s and Lewy body dementia
The researchers are working with two groups of patients: those with Alzheimer’s and Lewy body dementia, in which abnormal protein deposits—called Lewy bodies—are present in the brain. These deposits affect and change the chemicals in the brain associated with thinking, movement, behavior, and mood. Lewy body and Alzheimer’s are similar, but have different symptoms.
“Lewy body dementia has different mechanisms in the brain, but it’s very commonly misdiagnosed as Alzheimer’s,” Bale says. “One of our goals is to separate people with Lewy body dementia from people with Alzheimer’s, because you need to treat them very differently. If you’re being misdiagnosed, the treatment is not going to work.”
The study includes people with mild cognitive impairment, known as the prodromal stage, which is “early dementia, before it’s full-blown,” Bale notes. “We’re looking at people who are likely to develop Alzheimer’s or Lewy body dementia, based on their family history and other factors.”
The aim of studying these groups is to detect symptoms earlier because existing research shows the earlier dementia is detected and diagnosed, the more likely doctors can treat or at least manage the symptoms.
The Cambridge-led study also involves another test group: healthy seniors. Bale says the team compares results from this control group to those from people with Alzheimer’s and Lewy body. “This is a really interesting group because we can try to see what healthy aging in brain function looks like,” she says. “We know there is some deterioration, but is it just forgetfulness or something more?”
Putting on a thinking cap
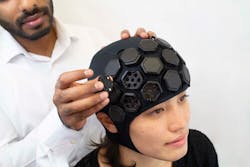
A second study involves a wearable device called LUMO, which features portable, high-density functional NIR imaging developed by optical neuroimaging R&D company Gowerlabs, a spinoff from a lab Bale was once involved with at University College London. The device resembles a swimming cap embedded with modular tiles that contain NIR light sources and detectors (see Fig. 1).
“We get the subjects to do lots of different cognitive tests so we can try to see how well their memory is working,” Bale says. “We can test different parts of the brain using this one device.”
For example, by positioning the tiles on the front of the device on the subject’s forehead, over the brain’s frontal lobe, the researchers can analyze the results as the subject performs various memory tasks. Placing the tiles over the brain’s motor cortex, which controls movement, they view the data as the subject walks, stands, sits, etc. Putting the tiles over the back of the head, where the visual cortex is, they gather information as the subjects view and discuss photos and other objects.
Unlike magnetic resonance imaging (MRI) and computed tomography (CT) imaging techniques, which require a patient to be still, NIR spectroscopy via the LUMO device monitors the brain as it functions naturally—allowing a much better view and understanding of how dementia impacts it.
NIR spectroscopy provides a huge advantage
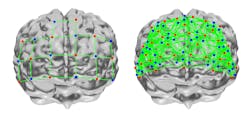
Using NIR spectroscopy imaging via the portable LUMO device not only offers more comprehensive views, but also is a more practical imaging method than conventional approaches (see Fig. 2).
“One of the biggest advantages is that it enables brain monitoring to be done anywhere,” Bale says. “When someone has dementia, getting them to come into a hospital for tests can be very challenging. And getting them to come into the hospital for a university research study is next to impossible, but with this device, we’re able to take the kit to people’s homes. We can monitor them doing natural tasks.”
This capability is very beneficial, particularly in the study of Lewy body dementia, which commonly presents motor function and movement problems.
“If you’re trying to put someone in an MRI scan, you have to tell them to lay out and be very still, so you’re not going to ever see natural motor function or natural movement,” Bale says. “But with our approach, we’re able to capture, for example, what your brain actually looks like when you’re trying to walk across your living room.”
Early results show promise
“We’re just starting to look at the dementia-diseased versus healthy brain, so we’re still in the very, very early stages,” Bale says. “We are seeing differences, but I can’t tell if it's significant yet because we’re probably seeing differences between people, not necessarily the dementia or Alzheimer’s. We don’t know just yet.”
A Ph.D. student and neuroscientist working with Bale has developed protocols for the research team and collected 15 datasets over several months. These results have already shown some differences between a person with dementia and one who is healthy, Bale notes. However, the team continues to work toward perfecting its approach to ensure it can make accurate diagnoses and differentiate between the various types of dementia. For instance, in the mild cognitive impairment groups, the researchers are still discovering differences between brains exhibiting signs of potential dementia and healthy brains that could someday help detect dementia earlier.
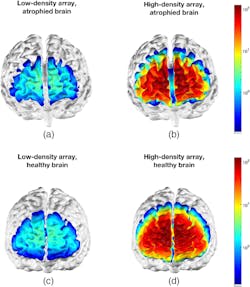
Their work has also shown that brain scans showing higher-density NIR spectroscopy arrays are significantly more sensitive to brain atrophy than traditional imaging methods (see Fig. 3).
“What has been shown definitively in the animal models and human trials, as well, is once dementia is really set in, your brain has literally already shrunk,” Bale says. “It's really hard to restore function, but if you can detect it early and prevent the deterioration, it’s going to be really important.”
While early detection is crucial, it’s unclear what treatments to pursue upon early diagnosis. How do you prevent the disease progression? Can it be treated even before symptoms present themselves? “At the same time,” Bale says, “people are constantly developing new therapies and medicines. We need to be ready with the diagnosis in case someone says, ‘Okay, we can stop it if we catch it 10 years earlier.’ Then we can provide our information from the NIR spectroscopy approach, which can go hand in hand with the therapy.”
About the Author
Justine Murphy
Multimedia Director, Digital Infrastructure
Justine Murphy is the multimedia director for Endeavor Business Media's Digital Infrastructure Group. She is a multiple award-winning writer and editor with more 20 years of experience in newspaper publishing as well as public relations, marketing, and communications. For nearly 10 years, she has covered all facets of the optics and photonics industry as an editor, writer, web news anchor, and podcast host for an internationally reaching magazine publishing company. Her work has earned accolades from the New England Press Association as well as the SIIA/Jesse H. Neal Awards. She received a B.A. from the Massachusetts College of Liberal Arts.