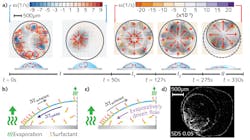
To understand why a dried drop of whiskey leaves a coating with relatively uniform thickness compared to the nonuniform "coffee-ring" stain left by other water-alcohol mixtures, researchers at Princeton University (NJ) used particle image velocimetry to compare fluid flows and particle deposition processes of evaporating liquids, which suggests a new coating method.
To visualize flows, 1-μm-diameter fluorescent polystyrene particles were added to whiskey, revealing multiple vortices, circulatory flows, and radial outward flows. Observing water-ethanol mixtures during evaporation did not show these circulatory flows, leading the researchers to posit that various chemical compounds within whiskey (most likely phospholipids from raw grains) are acting as surfactants—compounds that lower the surface tension within a droplet. Further analysis showed a nonuniform dried pattern and the realization that yet another compound was also playing a role in particle deposition from the whiskey mixture. So the researchers developed an ethanol-water-surfactant-polymer "model liquid" that mimics whiskey upon evaporation, which can be applied to developing uniform coatings via evaporative processes. Reference: H. Kim et al., Phys. Rev. Lett., 116, 124501 (March 24, 2016).
About the Author

Gail Overton
Senior Editor (2004-2020)
Gail has more than 30 years of engineering, marketing, product management, and editorial experience in the photonics and optical communications industry. Before joining the staff at Laser Focus World in 2004, she held many product management and product marketing roles in the fiber-optics industry, most notably at Hughes (El Segundo, CA), GTE Labs (Waltham, MA), Corning (Corning, NY), Photon Kinetics (Beaverton, OR), and Newport Corporation (Irvine, CA). During her marketing career, Gail published articles in WDM Solutions and Sensors magazine and traveled internationally to conduct product and sales training. Gail received her BS degree in physics, with an emphasis in optics, from San Diego State University in San Diego, CA in May 1986.