Biomedical Imaging: Squeezed light brings quantum-enhanced contrast and resolution to bioimaging
Quantum correlations between photons can be used to surpass shot-noise and diffraction limits in biological-microscopy applications. Because biological subcellular structures have a physical size far below the diffraction limit of visible light, quantum-enhanced imaging methods can enable sub-diffraction-limited imaging.
Unfortunately, while proof-of-principle quantum-enhanced experiments have shown imaging improvements using entangled photons, power levels from entangled-photon sources are low, and such experiments also remain constrained by the diffraction limit. An alternative quantum-enhanced method developed by researchers at the University of Queensland (Brisbane, Australia) and Australian National University (Canberra, Australia) instead uses squeezed states of light to achieve sub-diffraction-limited imaging of biological structures.1, 2
Generating squeezed light
Squeezed light is a nonclassical state of light wherein the noise of one of its components (the amplitude or the phase) is below the standard quantum limit; that is, the photocurrent measured for the light is exceedingly low—much lower than the noise expected from classical photons. And unlike entangled photon sources, squeezed light can be generated with a light source of any power level to improve imaging contrast.
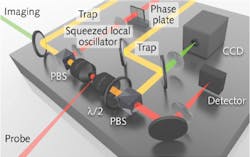
To generate squeezed light in an experimental setup that tracks particles within a yeast cell, an amplitude-squeezed local oscillator and an amplitude-modulated probe that provides dark-field illumination track the particle via the interference signal between probe-scattered light and local-oscillator light (see figure). The real-time viscoelasticity measurements of particles within the living yeast cells—which were gathered at a rate 64% faster than using coherent laser light—reveals information about the structure and function of the cells and can even be used to discriminate between healthy and cancerous cells.
Photonic-force microscopy
Beyond viscoelasticity measurements, squeezed light can also be used to track how the thermal vibrations of a nanoparticle influence surrounding particles, providing local information about subcellular structures as small as 10 nm. In a process called quantum-enhanced photonic-force microscopy (PFM), the nanoparticle’s interaction with other structures in the cell functions much as a cantilever tip does when it scans an object via atomic-force microscopy (AFM) imaging methods. As the nanoparticle explores its environment, subcellular structures measurably influence its thermal vibrations. By mapping out the profile of the thermal motion at different positions, a full spatial profile can be constructed.
So far, the squeezed-light research only provides physical information via particle tracking in one dimension. This prevents the reconstruction of full images, although it allows a device characterization that demonstrates a 14% improvement in resolution compared to traditional coherent laser light imaging. “This provided a clear look at the potential of our technique, and the steps we need to take to make it truly competitive,” says Michael Taylor, postdoctoral researcher at the University of Queensland. “We have improved the design and are working to develop three-dimensional [3D] imaging capability; if successful, 3D images with subnanometer resolution could be possible—an unprecedented capability.”
REFERENCES
1. Michael Taylor et al., “Squeezed light pushes the quantum limit in biological microscopy,” SPIE Newsroom online (June 29, 2014); doi:10.1117/2.1201406.005506.
2. Michael Taylor et al., Nat. Photonics 7, 229–233 (2013).
About the Author

Gail Overton
Senior Editor (2004-2020)
Gail has more than 30 years of engineering, marketing, product management, and editorial experience in the photonics and optical communications industry. Before joining the staff at Laser Focus World in 2004, she held many product management and product marketing roles in the fiber-optics industry, most notably at Hughes (El Segundo, CA), GTE Labs (Waltham, MA), Corning (Corning, NY), Photon Kinetics (Beaverton, OR), and Newport Corporation (Irvine, CA). During her marketing career, Gail published articles in WDM Solutions and Sensors magazine and traveled internationally to conduct product and sales training. Gail received her BS degree in physics, with an emphasis in optics, from San Diego State University in San Diego, CA in May 1986.