SPECTROSCOPY: Quantum-cascade-laser spectrometer measures gases in atmosphere
The ability to measure the gases that contribute to global warming, such as methane (CH4), carbon dioxide (CO2), chlorofluorocarbons (CFCs), and nitrous oxide (N2O), is important for long-term tracking of emissions and how they affect the Earth’s atmosphere.1 Infrared-laser absorption spectroscopy is an effective tool for measuring these molecular trace gases down to 1 in 1012 part by volume levels. But the method is limited by the lack of tunable sources in the region of the fundamental vibrational absorption bands. Now, scientists in France have developed a single-mode tunable distributed-feedback quantum-cascade-laser (QCL) spectrometer for use in the mid-infrared region at room temperatureuseful for isolation of CH4 and N2O lines around 1270 cm-1 (7.9 µm).
Distributed-feedback QCLs work well as spectroscopic sources because of their narrow linewidth, high output power of more than 10 mW, reliability, and compactness. Recently, QCLs expanded into room-temperature operation, adding to their practical potential for incorporation into portable, in-field systems. Atmospheric scientists at the Université de Reims (Reims, France) chose a QCL from Alpes Lasers operable between −30°C and +30°C as the source for their optical platform. The laser is tunable from 1265 to 1274 cm-1, providing approximately 30 mW of power at a maximum current of 550 mA.
To create the spectrometer system, the laser beam was split into one part coupled with a Fabry-Pérot germanium etalon for relative frequency calibration and one part focused with an f = 1 m spherical mirror at the entrance to the absorption cell. Both beams were then focused by an off-axis parabola onto two liquid-nitrogen-cooled mercury cadmium telluride detectors. The multiple reflection-absorption cell consisted of three spherical mirrors similar in principle to a White cell but with multiplier behavior and thus the optomechanical stability of a Herriott cell, creating a long optical path of 140 m total. Sample atmospheric gas was collected outside the laboratory and pressurized to 40 mbar across the inlet orifice of the system to reduce pressure broadening of the absorption lines of the gas species.
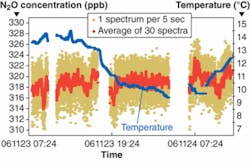
The researchers used the system to detect time-dependant fluxes of N2O in soil, which is a byproduct of nitrogen fertilizer when it is irrigated, and other greenhouse gases, such as CH4. After monitoring concentrations of N2O and CH4 for 26 hours, the system measured a mean N2O volume mixing ratio of 319 parts per billion and a mean CH4 volume mixing ratio of 1.93 parts per million, typical of background troposphere boundary layer levels (see figure). The system was able to discern no cycling of the levels of these gases, and it ruled out any effect of weather on the concentrations of the gases at ground level over the measurement period, demonstrating the feasibility of a QCL spectrometer for atmospheric gas monitoring.
Further iterations of the system currently under development will include reduction of the cell volume and the use of room-temperature detectors. This will permit better control of temperature, pressure, and flow, says team member Virginie Zéninari. “Increasing the stability and precision of the instrument will enable in situ measurements over several weeks,” she said. In several regional projects with colleagues from the French National Institute on Agronomical Research, the Reims team plans to use the improved version of the spectrometer to produce in situ measurements of N2O, for example, due to fertilizers in vine fields. Longer term, the hope is that such a powerful spectrometer will find more widespread use in the agronomical and biogeochemical communities.
REFERENCE
1. V. Zéninari et al., Appl. Opt. 47 (9), 1206 (March 2008).
About the Author
Valerie Coffey-Rosich
Contributing Editor
Valerie Coffey-Rosich is a freelance science and technology writer and editor and a contributing editor for Laser Focus World; she previously served as an Associate Technical Editor (2000-2003) and a Senior Technical Editor (2007-2008) for Laser Focus World.
Valerie holds a BS in physics from the University of Nevada, Reno, and an MA in astronomy from Boston University. She specializes in editing and writing about optics, photonics, astronomy, and physics in academic, reference, and business-to-business publications. In addition to Laser Focus World, her work has appeared online and in print for clients such as the American Institute of Physics, American Heritage Dictionary, BioPhotonics, Encyclopedia Britannica, EuroPhotonics, the Optical Society of America, Photonics Focus, Photonics Spectra, Sky & Telescope, and many others. She is based in Palm Springs, California.
